 And don’t forget to put the unit g/mol to your final calculated molar mass.First solve the brackets, then multiplications and at last do the final addition. OR (a) What type of deviation is shown by a mixture of (b) ethanol A solution and of acetone glucose (molar Give reason. Always follow the calculation order to avoid any mistakes in calculation.But all these units (i.e g/mol, grams/mole and g/mole) are the same. Transcribed Image Text: You are asked to prepare a 1.000L solution of 45 M CHOs (glucose, molar mass 180 16 g/mol) in a lab by dissolving 811.0gof glucose in water Consider the following two scenarios in which you commit a user emor while preparing this solution Prepared in a volumetric flask Prepared in a beaker Assumed volume 1. In some books, you may see the unit of molar mass as grams/mole or g/mole. Question: 40 g of glucose (Molar mass 180) is mixed with 200 mL of water.You may need the periodic table to calculate molar mass. I hope you have understood the short and simple calculation for finding the molar mass of Glucose. Molar mass is not to be mistaken with molecular weight the mass of a single molecule of a substance, given in daltons (e.g., a single HO particle is 18 u). Hence the Molar mass of Glucose (C6H12O6) is 180.156 g/mol. 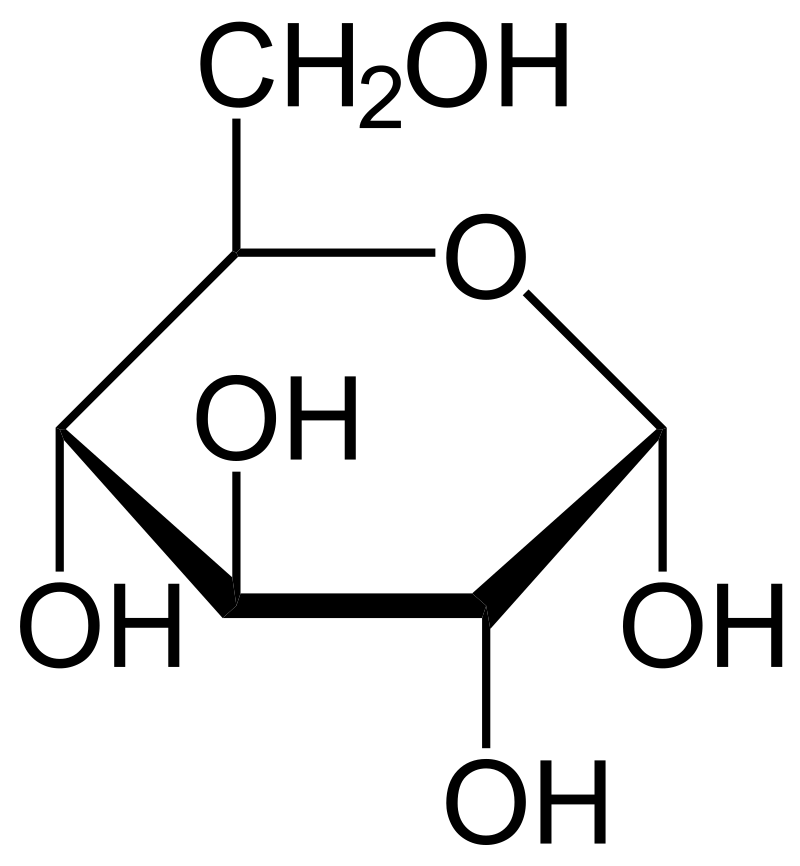
So, Molar mass of Glucose (C6H12O6) = Molar mass of 6 Carbon (C) atoms + Molar mass of 12 Hydrogen (H) atoms + Molar mass of 6 Oxygen (O) atoms. You can see that in Glucose (C6H12O6), there are 6 Carbon atoms, 12 Hydrogen atoms and 6 Oxygen atoms. Then you would have the same concentration of molecules in each that is, drop for drop, each solution would contain the same number of molecules.Now, to calculate the molar mass of Glucose, you just have to add the molar mass of all the individual atoms that are present in Glucose. To correct the problem, you should make the solution with the weights of sucrose and glucose in a ratio of 342:180. So a 35% solution of glucose would contain almost twice as many molecules as a 35% solution of sucrose. The sucrose molecule (MW = 342) is almost twice as heavy as the glucose molecule (MW = 180). The willingness of the honeybee to respond to the presence of sugar dissolved in water is dependent on the number of sugar molecules in a given volume of the solution. But there is a problem with this approach. When calculating molecular weight of a chemical compound, it tells us how many grams are in one. This would involve, in each case, dissolving 350 parts by weight (e.g., grams) of sugar in 650 parts (g) of water, thus producing 1000 g of each solution. Finding molar mass starts with units of grams per mole (g/mol). You might offer the bees the choice between, say, a 35% solution of sucrose (common table sugar) and a 35% solution of glucose (a natural component of honey). Molar mass is the mass of 1 mole of the solute. One way to do this would be to make up several different solutions and see which one the bees prefer to harvest. in the compound and convert Molecular formula and empirical formula of glucose. 
Why is it important to know the molecular weight of a compound?įor an example, let us assume that you want to study the response of honeybees to solutions of various kinds of sugars. Definitions of molecular mass, molecular weight, molar mass and molar. (We shall ignore the tiny error introduced by the presence of traces of other isotopes - 17O, 18O, and 2H among the predominant 1H and 16O atoms.) Thus the molecular weight ( MW) of water is 18 daltons. How many grams of glucose (molar mass 180.16 g mol 1) must be dissolved in 251 g of water to raise the boiling point to 106.99 C Mass of glucose i. /molecular-structure-of-glucose-85758435-5aeb1780a474be00361dff65.jpg)
The unit of weight is the dalton, one-twelfth the weight of an atom of 12C. The weight of a molecule is the sum of the weights of the atoms of which it is made. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
